Peptide-Fe complexes have been identified as promising iron-chelators[1], and peptide segments that change their conformation due to external stimuli have been used in peptide-based molecular devices[2]. Additionally, experiments modeling volcanic or hydrothermal settings have shown that amino acids can be converted into their peptides by use of coprecipitated (Ni,Fe)S and CO[3]. Finally, an Integrated Peptide and Protein prediction Framework based on Fused features and Ensemble models (IPPF-FE) has been proposed to precisely predict peptides[4].
:max_bytes(150000):strip_icc():format(webp)/101121-vitamin-c-serum-lead-2000-fe6dff3877a9498baf198d958f108650.jpg)
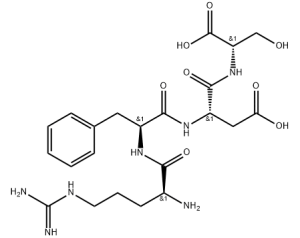
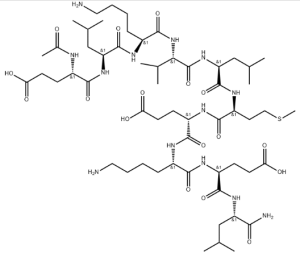
Peptide-Fe complexes are formed mainly through the carboxyl and hydroxyl oxygen bond[1]. Different metal ligands have been used to synthesize peptide-iron complexes[2], which have properties such as containing 5.6% Fe[3] and potential to reduce undesirable sensory changes in food products[4]. Gas phase studies of metal-peptide complexes can help understand intrinsic properties of the complex, such as 2H + Fe]+ ions[5].
Peptide-Fe complexes are generally stable, with studies showing that they can maintain their structural and physical-chemical stability over 1 year of storage[1]. The peptide-zinc complex has also been shown to enhance the stability of zinc during in vitro gastrointestinal digestion[2]. Additionally, a strong ionic signal corresponding to a FeIII complex was observed with LSIMS in the positive ion mode[3].
1
nih.gov
2
acs.org
3
science.org
4
oup.com
5
remetide.com